




1 / 5










| Still deciding? Get samples of $ ! US$ 60000/Set Order Sample |
The quality of effluent water meets the requirements of Chinese Pharmacopoeia (Cp2020), U.S. Pharmacopoeia (USP43), and European Pharmacopoeia (EP10) for purified water or high purity water depending on system configurations.

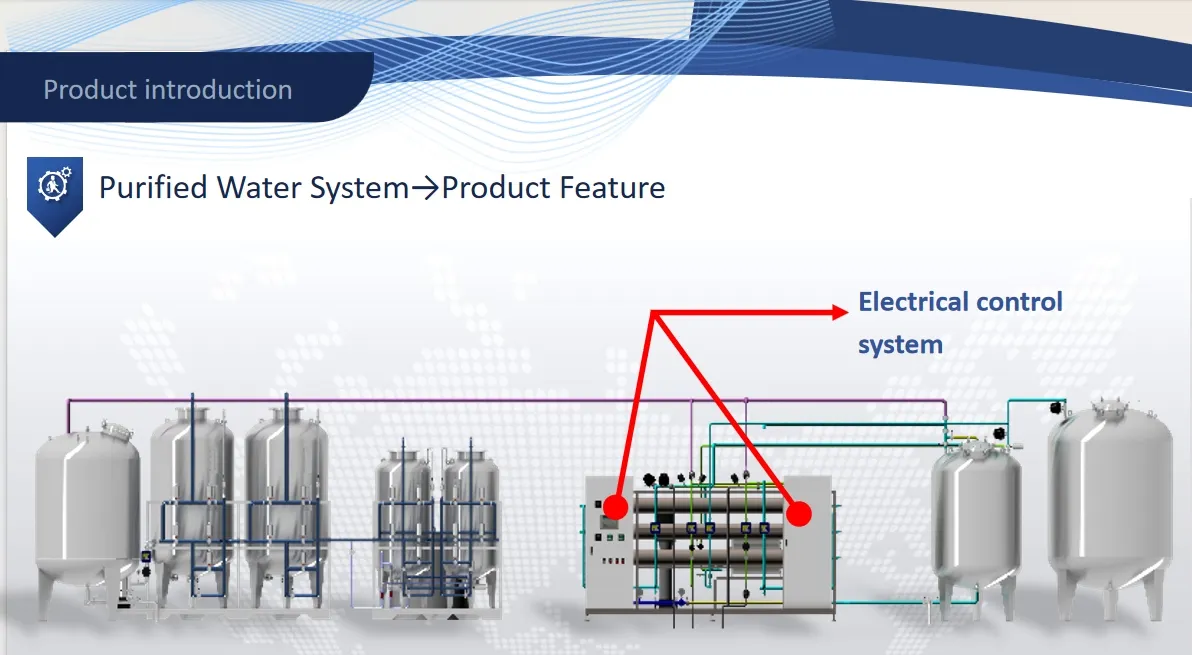
The storage tank automatically controls water inlet via a PLC-managed electric slow-opened butterfly valve, preventing pressure impact. The pre-treatment unit includes mechanical filters, active carbon filters, and double softeners (A-B configuration).




| Capacity | 1500L/H |
| Utility Requirements | Power, Electric, Industrial Steam |


Why Partner With Us?
Complementary Solutions:
 Liftora Arm
Liftora Arm